INJECTOR PRODUCTION INFORMATION

SYRINGE PRODUCTION LINE

Syringe Manufacturing
According to research by TTT World, the annual global usage of syringes exceeds 16 billion. As a key player in this field, TTT World manufactures vaccine syringes and a wide range of medical syringes that comply with international safety and performance standards.
What is a Syringe?
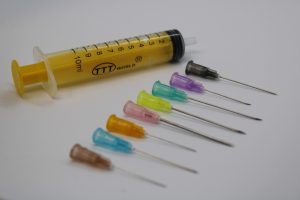
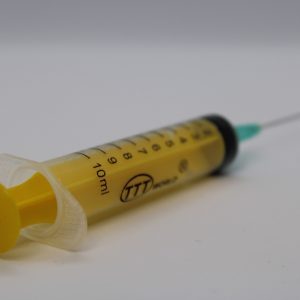
A syringe is a small pump used for drawing in and expelling liquids or air. In medical contexts, it is used to inject fluids into tissues or cavities. A standard syringe consists of a cylindrical barrel, a plunger, and a Luer tip compliant with ISO 80369-7, compatible with hypodermic needles.
Product Range and Technical Specifications
- Available in two-piece and three-piece models
- Can be supplied with or without needles
- Produced from biocompatible, non-toxic, and pyrogen-free materials compliant with ISO 7886-1
- Sterilized using EO (ethylene oxide)
- Packaged in blister packs made of medical-grade paper and transparent film
- 100% leak-proof with laser-cut hypodermic needles made from medical-grade stainless steel, certified under ISO 7864
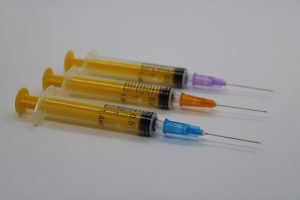
TTT World offers syringes in multiple colors, sizes (mL/cc), needle gauges, and intended uses. The materials include medical-grade polypropylene, latex-free rubber, and precision-molded pistons with gaskets. Syringes are sterilized post-production and are available in single-use, non-reusable configurations.
Indications and Contraindications
Indications: Single-use syringes are intended for delivering medication via various injection routes based on the properties of the drug.
Contraindications: Do not use if the needle or packaging is damaged, or if the expiration date has passed. Syringes must not be reused, and protective caps should never be removed by mouth.
Syringe Types Available
- Sterile syringes with or without needles
- Individually packed hypodermic needles
- Available in various volumes and gauge sizes
Quality Management and Production Process
The entire manufacturing and quality control process is governed by TTT World’s Quality Management System (QMS). All incoming raw materials are stored in designated quarantine areas until they pass quality checks:
- Accepted materials are moved to the green-tagged acceptance zone
- Rejected materials are returned and placed in the red-tagged rejection area
Materials like PP/PE granules are injected into molds to form barrels and plungers. Printed components go through serigraphy machines, while assembled parts (barrel, plunger, gasket) are processed using automated assembly and sealing systems.
Blister packaging involves heat-sealing medical-grade paper with plastic film. Once packaged, all syringes undergo EO sterilization before being packed into inner and outer cartons and stored for shipment.
Global Reach, Local Excellence
TTT World’s commitment to quality and performance has earned it top search rankings as a trusted syringe manufacturer in Turkey. Our products meet the highest hygiene and safety standards and are exported worldwide.
For product inquiries or pricing of 1cc syringes, vaccine injectors, or needle types, please contact us directly.
All syringes and needles are:
- Sterile
- Non-toxic
- Pyrogen-free
Explore our full Blood Collection Tube Product Line or read more about ETO Sterilization Services on our website.